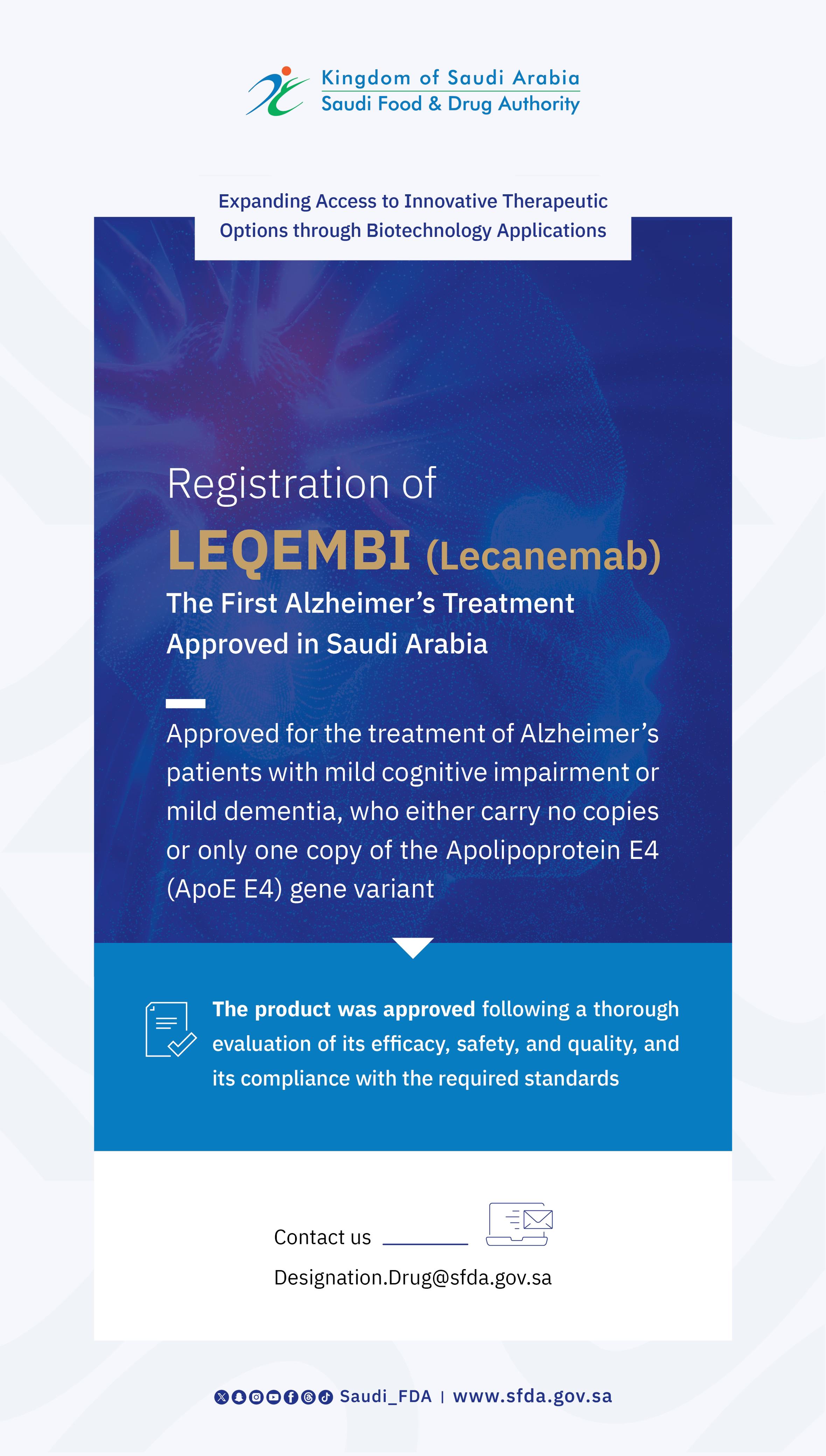
SFDA Grants Breakthrough Designation to "Bemlayo" for Adults with Obesity or Overweight
2026-03-10
The Saudi Food and Drug Authority (SFDA) has granted Eli Lilly Breakthrough designation to Bemlayo (Orforglipron), which is indicated as an adjunct to healthy diet and physical activity to reduce excess body weight and maintain weight reduction long term in adults with obesity or adults with overweight in the presence of at least one weight-related comorbid condition.
Registration is Subjected to Full Scientific Evaluation
The SFDA emphasizes that this designation allows Bemlayo to be submitted and reviewed under the Breakthrough Track, subject to specific regulatory controls and mechanisms. This designation does not constitute a marketing authorization of the drug in Saudi Arabia. A final decision regarding registration will be announced following a full technical and scientific evaluation of the complete registration dossier.
The SFDA Breakthrough Medicine Program Expedites Access to Targeted Treatments
The Program aims to expedite access to innovative and effective treatments for serious diseases where existing therapies may be inadequate. To qualify for this designation, a drug must have completed clinical trials demonstrating promising efficacy and safety, offer a significant therapeutic advantage over current options, target serious or life-threatening conditions, and exhibit a positive benefit-risk balance. Notably, the product should not be registered with any other regulatory authority at the time of application.
For further information about the Breakthrough Medicines Program, please refer to the guide available on the SFDA website: