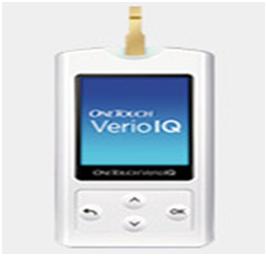
Saudi Food and Drug Authority warns from using OneTouch® Verio® IQ blood glucose meter (home use)
2015-05-05
تود الهيئة العامة للغذاء والدواء ممثلة بقطاع الأجهزة و المنتجات الطبية أن تحذر المستهلك من استخدام جهاز قياس نسبة السكر في الدم طراز ون تتش فريو أي كيو (OneTouch® Verio® IQ) للاستخدام المنزلي والذي يصنع من قبل شركة لايف سكان جونسون آند جونسون (Lifescan Johnson & Johnson Company)
حيث علمت الهيئة من خلال الشركة المصنعة للجهاز عن قيامها بسحبه واستبداله وذلك للأسباب التالية: